FibroScan® Solutions in MASH* Clinical Trials
FibroScan® and its associated solutions can be leveraged to significantly reduce screen failure rate, accelerate patient enrollment, and monitor therapeutic response.
Download the White Paper and enjoy the reading!
*Formerly known as NASH

Our solutions can support the development of MASH therapies
with the reference liver non-invasive point-of-care testing solutions
- Accelerating drug development,
- De-risking trial execution
- Preparing for commercialization
Overview of the white paper
Schematic of a typical Phase 3/4 clinical trial for accelerated drug approval
to treat patients with non-cirrhotic MASH.
(EOT: End of Treatment, M: Month)
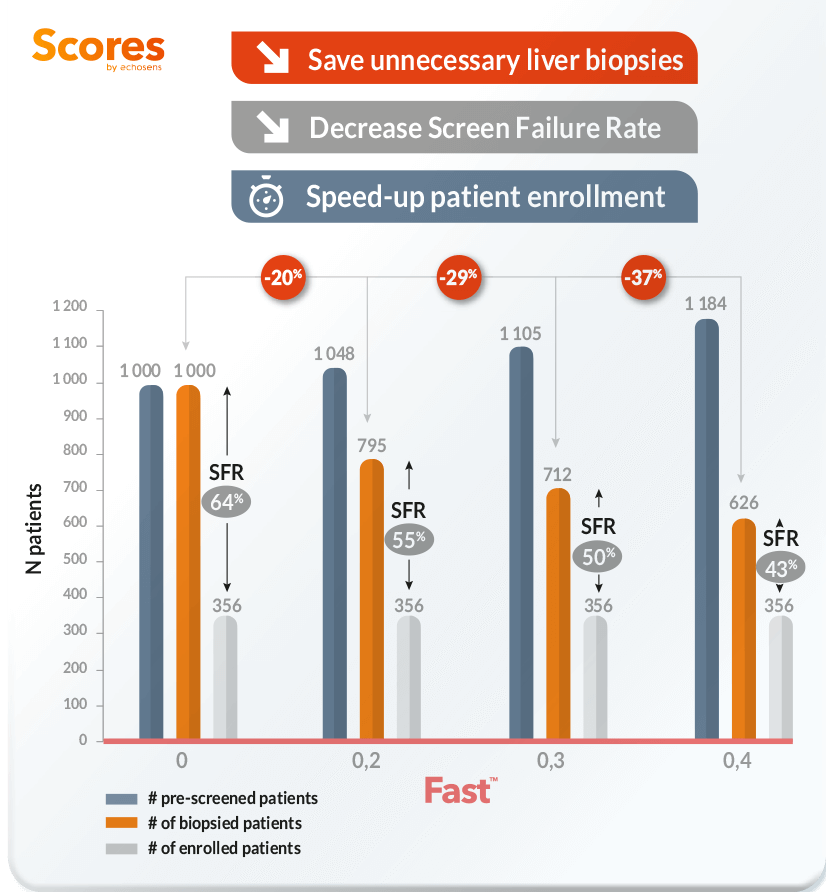
Impact of the use of Fast on patient enrollement
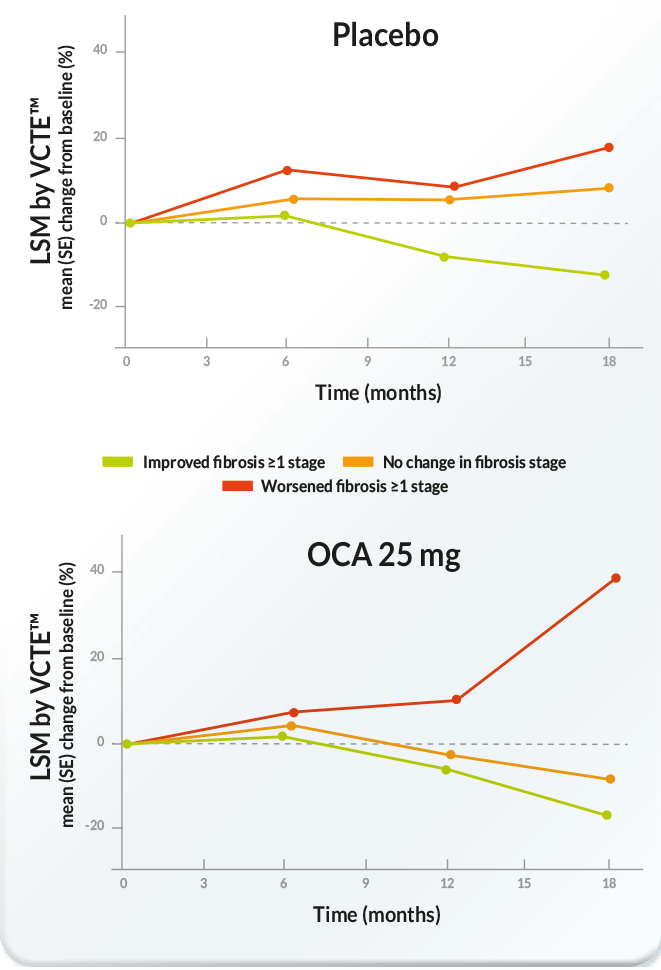
Mean percentage change from baseline in LSM by VCTE™ over time
by treatment group and histological fibrosis improvement status
(adapted from [1])
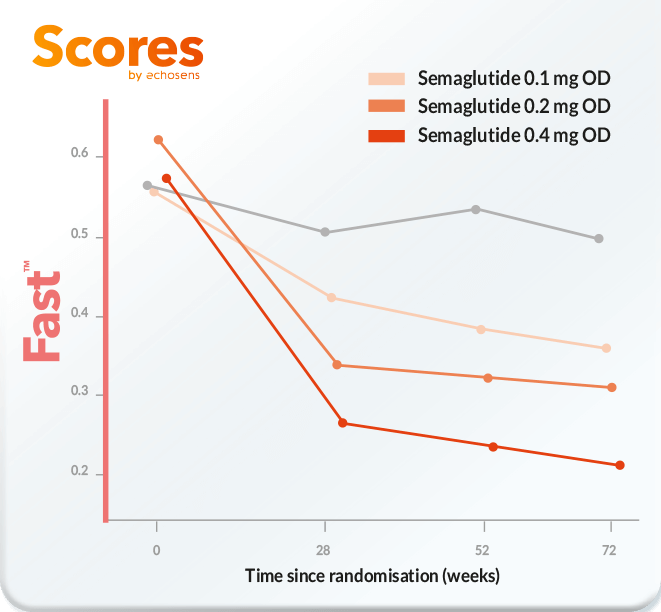
Change from baseline to week 72 of Fast™ score
Subcutaneous Semaglutide Phase 2b trial
(adapted from [2])
[1] – Rinella M, Dufour J-F, Anstee QM, Goodman Z, Younossi Z, Harrison SA et al. Non-invasive evaluation of response to obeticholic acid in patients with MASH: Results from the REGENERATE study. Journal of Hepatology 2021.
[2] – Wong VW-S, Quentin A, Geerts A, Mette K, Ladelund S, Ratziu V et al. Change in FibroScan-aspartate aminotransferase (FAST) score is associated with histological improvement in non-alcoholic steatohepatitis activity: The Journal of Hepatology; 2021.
Products in the FibroScan® range are class IIa medical devices according to Rule 10 of ANNEX VIII of Regulation EU 2017/745 (CE 0459) and are manufactured by Echosens™. FibroScan® devices are intended to provide: Liver stiffness measurements at a shear wave frequency of 50 Hz, liver ultrasound attenuation measurements (CAP: Controlled Attenuation Parameter) at 3.5 MHz and spleen stiffness measurements at a shear wave frequency of 100 Hz (FibroScan® 630 Expert only). These non-invasive devices are intended to aid clinical management, diagnosis, and monitoring of patients with confirmed or suspected chronic liver disease, as part of an overall assessment of the liver. FibroScan® devices may aid the healthcare professionals in the assessment of liver fibrosis, steatosis, and in determining the likelihood of cirrhosis, and its complications (FibroScan® 630 Expert only). They are used, in conjunction with other clinical and laboratory data, during liver assessment in patients with confirmed or suspected chronic liver disease. Examinations with FibroScan® devices shall be performed by an operator who has been certified by the manufacturer or its approved local representative. Operators are expressly recommended to carefully read the instructions given in the user manual and on the labelling of these products. Check cost defrayal conditions with paying bodies.