FibroScan® is a non-invasive solution for comprehensive management of liver health. It is a painless, reimbursable exam that provides immediate results at the point-of-care. FibroScan® can be performed by a trained operator (physician, advanced practice provider, nurse, sonographer, medical assistant) and provides standardized examinations with precision and reproducibility that can be utilized in 99% of patients. It is equipped with patented technology and proprietary algorithms to deliver consistently accurate results.
The FibroScan® device (Models: 502 Touch, 530 Compact, 430 Mini+, 630 Expert and FibroScan GO/230) is intended to measure liver stiffness using Vibration-Controlled Transient Elastography (VCTE®) at 50 Hz shear wave frequency and liver ultrasound attenuation coefficient (CAP) at 3.5 MHz. FibroScan® liver stiffness measurements (LSM) by VCTE® may aid the physician in determining the likelihood of cirrhosis and may be used as an aid in the assessment of liver fibrosis. FibroScan® CAP measurements may be used as an aid in the assessment of hepatic steatosis.
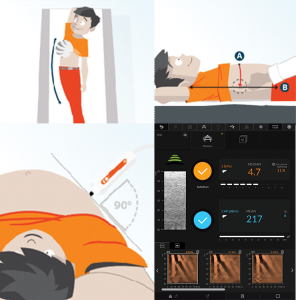
The exam is performed in an outpatient setting and typically takes about 5–10 minutes. During the exam, the patient lies on their back with their right arm raised behind their head to allow access to the liver area. A trained operator places the FibroScan® probe on the skin along the right side of the rib cage. Using ultrasound image guidance, the operator identifies liver tissue and ensures the region of interest is clear of structures that could affect the measurement.
The device then delivers a series of gentle vibrations, creating shear waves that pass through the liver. Ultrasound technology measures how fast these waves travel, which correlates with liver stiffness. At the same time, the system assesses ultrasound attenuation to estimate liver fat content (CAP). The exam captures multiple measurements and images, which are reviewed by the provider to confirm accuracy and interpret results.
Results are available immediately and can be used to support clinical decision-making, monitor disease progression, and guide patient management.
CPT® Code for FibroScan®
76981 Ultrasound, elastography; parenchyma (eg, organ)
CPT® 76981 is appropriate for a FibroScan® exam and interpretation. Per the American College of Radiology’s (ACR) 2025 Ultrasound Coding User’s Guide, 76981 describes ultrasound elastography (USE), a noninvasive imaging method utilized to assess and visualize the relative stiffness of tissues (shear wave). The primary clinical application of USE is in the evaluation of chronic liver disease. When imaging/image guidance is used to perform USE on any organ, CPT® code 76981 should be utilized. Per the American Medical Association, the imaging component of this code is not for a diagnostic ultrasound image, but it rather encapsulates the ultrasound image component of this exam. FibroScan® utilizes ultrasound for image guidance and archives liver images on the exam report for interpretation. Therefore, FibroScan® is consistent with the imaging component described in 76981.
Distinction Between 76981 and 91200
The primary difference between CPT® codes 91200 and 76981 is the use of ultrasound imaging/image guidance. If it was not used, or if the work was not completed (images were not interpreted by the provider, or archived in the medical record), then 91200 should be billed for liver elastography.
FAQs
Is B-mode ultrasound a requirement for CPT® 76981?
No, B-mode is NOT a requirement for CPT® 76981. There is no specific ultrasound imaging modality requirement (B-mode, 1D, 2D, etc.) per the official guidance by ACR regarding utilization of the code. CPT® 76981 simply requires ultrasound imaging/guidance to be utilized, and that providers interpret the images as part of the exam.
How does FibroScan® use ultrasound?
FibroScan® uses ultrasound technology in two ways:
Ultrasound image guidance (USE)
Used for probe positioning and visual guidance to identify the region of interest (ROI), and to ensure the ROI is free and clear of structures/lesions that may impact shear wave measurements.
The images are stored and captured on the report and interpreted by the provider to ensure elastographic measurements were taken on a location of the liver free of structures (lung/diaphragm, etc.) that may affect the results of the exam.
Measure shear wave speed (VCTE®)
Once the shear wave is generated, ultrasound is used to measure the speed of the shear wave resulting in a shear wave image and a kPa value (correlate for fibrosis)
Other information online suggests that FibroScan® isn’t considered imaging because the FDA only classifies B-mode as imaging. Is this correct?
No, it is not correct.
The FDA is a regulatory agency with its own set of rules and classifications and it does consider FibroScan® to be an imaging device.
FibroScan® is considered a Diagnostic Ultrasound System by the FDA based on substantial equivalence to an Ultrasonic Pulsed Echo Imaging System and a Diagnostic Ultrasonic Transducer (Source: FDA 510(k) Summary K223902).
The American College of Radiology sets guidance for the 7000 series of CPT® codes and the 2026 ACR Coding Guide for Ultrasound provides specific guidance for each code in this series.
As mentioned above, the ACR has no guidance requiring the use of B-mode imaging for CPT® 76981.
What are best practices for documentation if utilizing 76981?
It is important to document the work performed during a FibroScan® exam in a clear and concise note and to save the exam report produced by the FibroScan® to the patient’s medical record.
The exam report satisfies the image archive component of the CPT® code.
Providers must review both the liver and shear wave images during the interpretation and note the findings in the note.
If this work is not performed, the note does not describe the interpretation, and the exam report is not saved to the medical record, it is not appropriate to bill 76981.
How to document FibroScan® and the imaging component
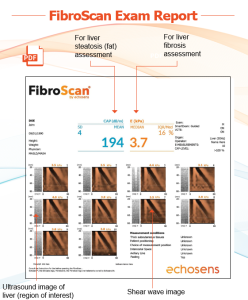
Upload the 1-page PDF FibroScan® exam report that includes the CAP and VCTE® values, along with 10 captured images to the patient’s EHR to be interpreted by the provider. Be sure to include a EHR note describing the FibroScan® exam (see example below).
Below is an EHR documentation template that also explains the utilization of imaging
The patient was placed in a supine position for five minutes prior to the start of the exam. Using the ____ (S, M, XL) probe, the ultrasound transducer was placed on the patient’s skin along the midaxillary line, and the center of the liver parenchyma was located using ultrasound imaging guidance. Once positioned, the transducer generated a series of no less than 10 transient vibrations producing elastic shear waves at 50 Hz. Each valid shear wave measurement produced a shear wave propagation map image, which was analyzed to confirm parallel shear wave margins, and a respective ultrasound image of the liver (region of interest) was captured. An equivalent liver stiffness measurement (LSM) value by vibration-controlled transient elastography (VCTE) was calculated in kPa. The corresponding ultrasound images were analyzed to confirm proper transducer positioning on the liver. Concomitantly, the transducer emitted a 3.5 MHz ultrasound signal to assess the liver ultrasound attenuation coefficient (CAP). All images were reviewed by the operator and the provider for technical accuracy. Data variability across the acquired measurements was quantified with IQR/ median ratio. The ______ (Physician/Nurse Practitioner/Physician Assistant) interpreted the examination results by reviewing the shear wave propagation map images, the corresponding ultrasound images, the quantity of images and measurements, and the IQR/median ratio. Comparison to threshold values for CAP and liver stiffness by VCTE was evaluated. The images have been archived and stored in the patient’s electronic medical record.
The median liver stiffness measurement (LSM) by VCTE was _____ kPa with an IQR/median ratio of ____.
The mean 3.5 MHz liver ultrasound attenuation coefficient (CAP) value was ____ dB/m.
Results demonstrate that the patient is (within/not within) threshold values and has an (increased/decreased) likelihood of _________.
Billing FAQs
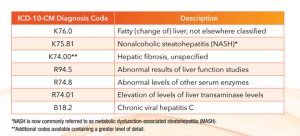
What ICD-10-CM diagnosis codes are used to bill for FibroScan®?
There are many diagnoses related to FibroScan®. The most common codes are listed below, however, it is advised to check with your payer and any applicable coverage policies regarding appropriate ICD-10-CM diagnosis code selection.
What are common modifiers related to use of FibroScan® and other services that may occur in during the same visit as a FibroScan® exam?
26: Professional component is outlined as a physician’s service, which may include technical supervision, interpretation of results, and a written report. May be appended to CPT® 76981 or 91200.
TC: Technical component includes the provision of all equipment, supplies, personnel, and costs related to the performance of the procedure. The payment for the technical component portion also includes the practice expense and the malpractice expense. May be appended to CPT® 76981 or 91200.
25: Used to indicate a patient’s condition required a significant, separately identifiable E/M service above and beyond what was associated with another procedure or service reported by the same physician or other qualified healthcare professional (QHP) on the same date of service. May only be appended to E/M service codes.
Coverage
FibroScan® is widely covered by Medicare and commercial payers, and most have written medical policies that explicitly state FibroScan® (VCTE®) is covered. Payers conduct comprehensive literature reviews to develop medical policy, and with nearly 6,000 peer-reviewed published papers, FibroScan® is the most studied elastography device on the market. Payers with medical policies that explicitly cover FibroScan® do not cover other elastography and consider them to be experimental/investigational due to a lack of published data.
The American Association for the Study of Liver (AASLD), recognize vibration-controlled transient elastography (VCTE), as used by FibroScan®, as a validated, noninvasive method to assess liver fibrosis and help guide patient management. Because FibroScan® has extensive clinical evidence and is considered the standard of care, payer policies commonly support coverage when medical necessity criteria are met.
Other elastography technologies may be reviewed separately by insurers and are typically labeled experimental or investigational if there is limited evidence, unclear diagnostic thresholds, or lack of guideline support for certain uses. Even if another device performs elastography, coverage is not interchangeable if the payer policy specifies a different modality.
Compliance and reimbursement risks when billing non-covered technologies
Billing CPT® 76981 for a modality that does not meet payer coverage criteria may create avoidable financial and compliance issues, including:
Claim denials if the payer determines the service was investigational or not medically necessary,
Patient financial liability when denied services are not covered benefits,
Audit risk if documentation does not support use of a covered modality,
Contractual risk related to inaccurate coding or misrepresentation of services performed,
Aligning the device used, clinical documentation, and payer policy requirements helps support appropriate reimbursement.
Additional Resources
American Society for Gastrointestinal Endoscopy 2024 Coding & Reimbursement Webinar