
Agile 3+
Identification of advanced fibrosis in MASLD patients.


Current MASH drug developments focus on patients with MASH at-risk of progression to cirrhosis or with compensated cirrhosis defined by Active Fibrotic MASH (MASH + NAS≥4 + F≥2).
Fast combines FibroScan® examination results (LSM by VCTE™ & CAP™) with an easily accessible blood biomarker (AST) to help identify patients with active fibrotic-MASH, at the point-of-care, reducing invasive and costly procedures.
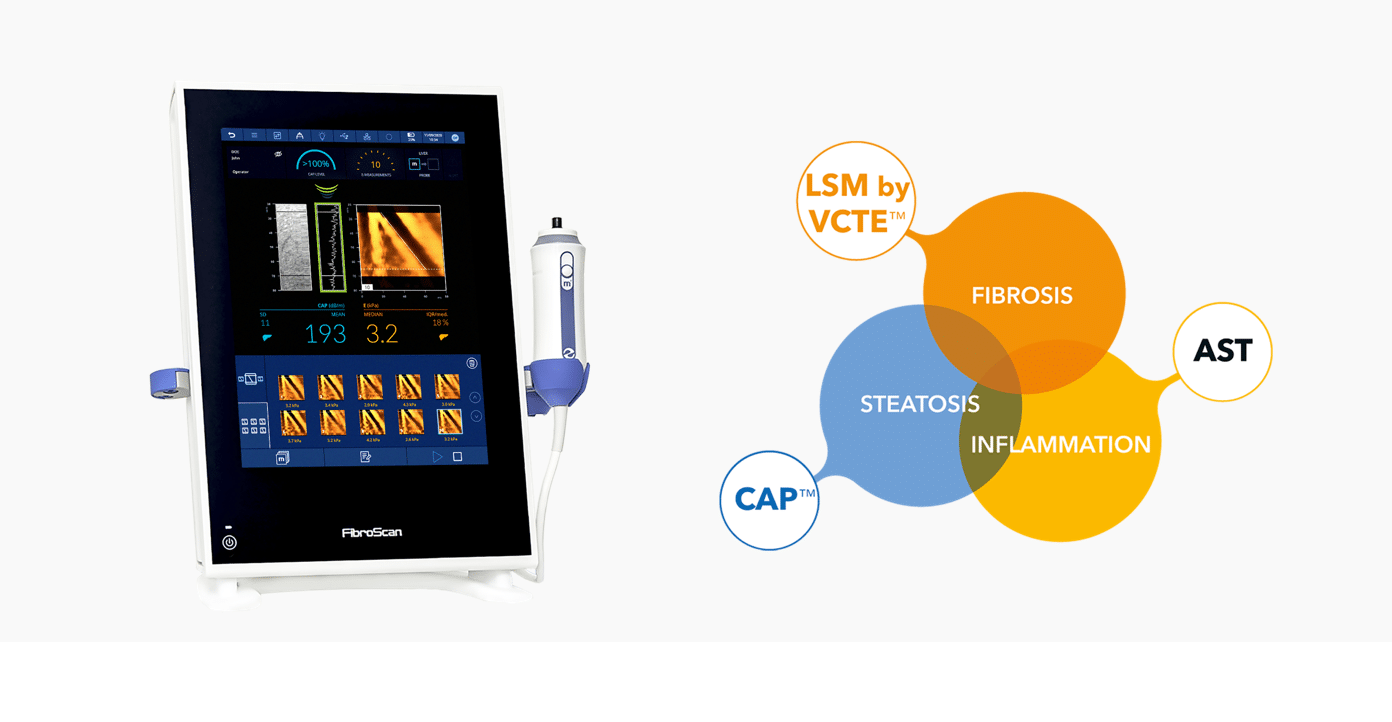
Good to excellent performance in derivation cohort as well as in external validation cohorts from different clinical settings (MASLD tertiary care unit, screening, bariatric surgery) and geographical origins (USA, Europe, Asia).
Several publications advocate the use of Fast.
Fast Score Calculator is a registered class 1 device (FDA). Fast Score Calculator is intended to compute the Fast score.
Fast Score Calculator was developed based on pools of cohorts and are published in peer-reviewed literature. It is taken from clinical literature and utilize proprietary FibroScan measurements in their execution. While these scores are about specific medical and health issues, Echosens does not represent them as substitutes for, or replacements of personalized medical advice, or to be used as the sole basis for making individualized medical or health-related decisions.
*MASLD/MASH is formerly knowed as NAFLD/NASH

Identification of advanced fibrosis in MASLD patients.

Identification of cirrhosis in patients with MASLD.

Cloud-based solution to assist clinicians in providing comprehensive liver care.

The non-invasive solution for comprehensive management of liver health