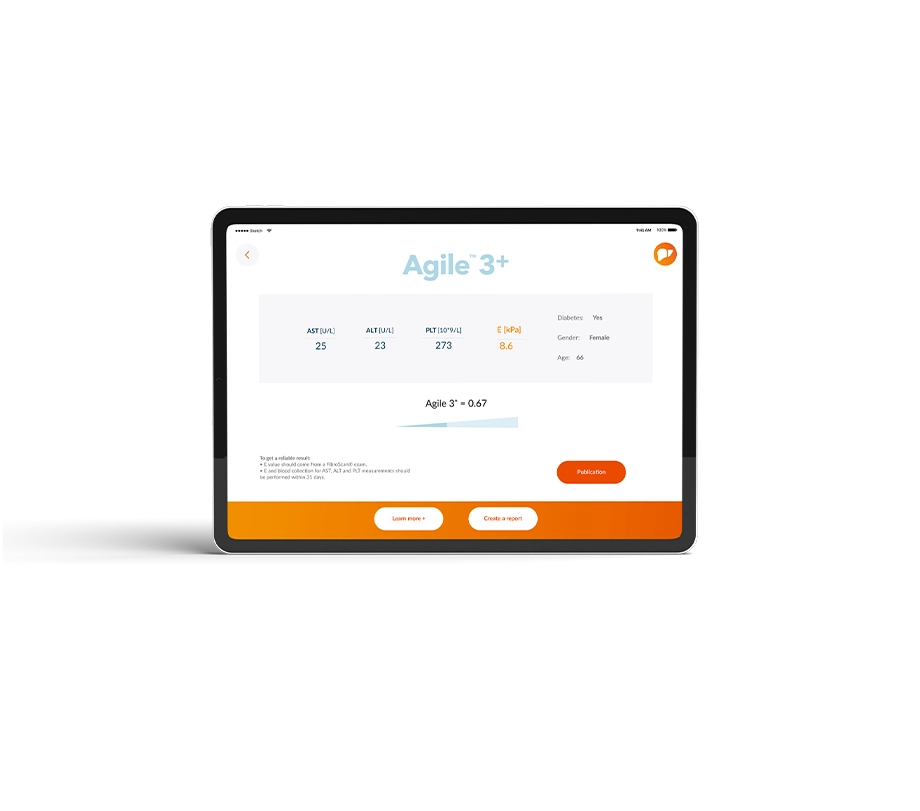
Agile 3+
Identification of advanced fibrosis in MASLD patients
Identification of cirrhosis in patients with MASLD
Agile 4 is a FibroScan®-based score


Cirrhosis is a leading cause of mortality worldwide.
Identifying cirrhosis early in patients helps to proactively manage the disease through screenings for liver-related complications such as hepatocellular carcinoma and esophageal varices.
Agile 4 combines LSM by VCTE™ with easily accessible blood biomarkers (AST, ALT, Platelets) to help in the identification of cirrhosis in MASLD patients to properly identify patients to start HCC and portal hypertension screening.

High clinical performances demonstrated in large external validation cohorts from different geographical origins.
Clinical validation: Sanyal et al., Enhanced diagnosis of advanced fibrosis and cirrhosis in individuals with MASLD using FibroScanbased Agile scores, Journal of Hepatology, 2022
Agile 4 is a class I medical device according to Directive 93/42/EEC. Agile 4 calculator is a tool for clinicians, computed from LSM (obtained from FibroScan® device), AST, ALT, platelets, diabetes status and gender, to aid in the identification of patients with suspicion of MASLD as having cirhhosis. It was developed based on a pool of retrospective cohorts and published in peer-reviewed literature. Agile 4 is presented as an educational service intended for licensed healthcare professionals. While this score is about specific medical and health issues, it is not a substitute for or a replacement of personalized medical advice and is not intended to be used as the sole basis for making individualized medical or health-related decisions.
*MASLD/MASH : formerly knowed as NAFLD/NASH
To access the Agile 4 User Guides, connect to myfibroscan.com:
Scores > Agile 4 > Learn more

Identification of advanced fibrosis in MASLD patients
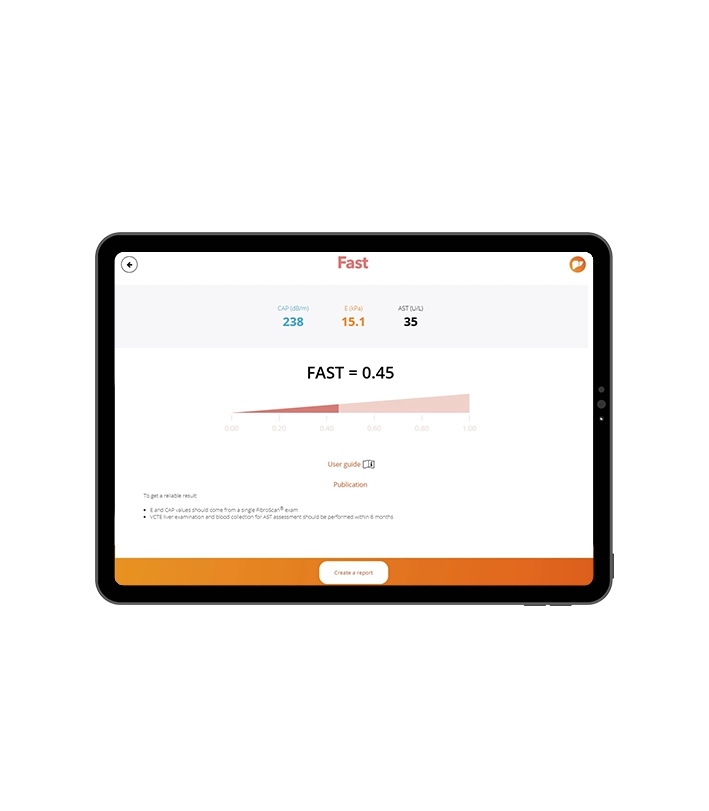
Identification of at-risk MASH patients

Cloud-based solution to assist clinicians in providing comprehensive liver care.

The non-invasive gold standard solution for comprehensive management of liver health