
Guided VCTE™
Seamless liver health assessment for all
The complete non-invasive solution for advanced liver disease management
Powered by unique, patented and validated parameters: LSM by VCTE™, CAP™ and SSM by VCTE™ as part of an overall assessment of the liver.


The spleen stiffness measurement (SSM by VCTE™) enables risk stratification of patients with advanced chronic liver disease and portal hypertension. 1, 2, 3
The embedded ultrasound guidance eases both liver and spleen targeting.
Assess and Monitor Portal Hypertension*
SSM by VCTE™ helps to assess and monitor portal hypertension, the main driver of cirrhosis, in a quick and non-invasive way.
Helps to Assess the Presence of Esophageal Varices*
SSM by VCTE™ can provide added value to help identify high-risk varices and prioritize endoscopies.
Determine Surgery*
SSM by VCTE™ has the potential to triage patients in the general surgical population by assessing portal hypertension, a known risk factor, prior to surgery.
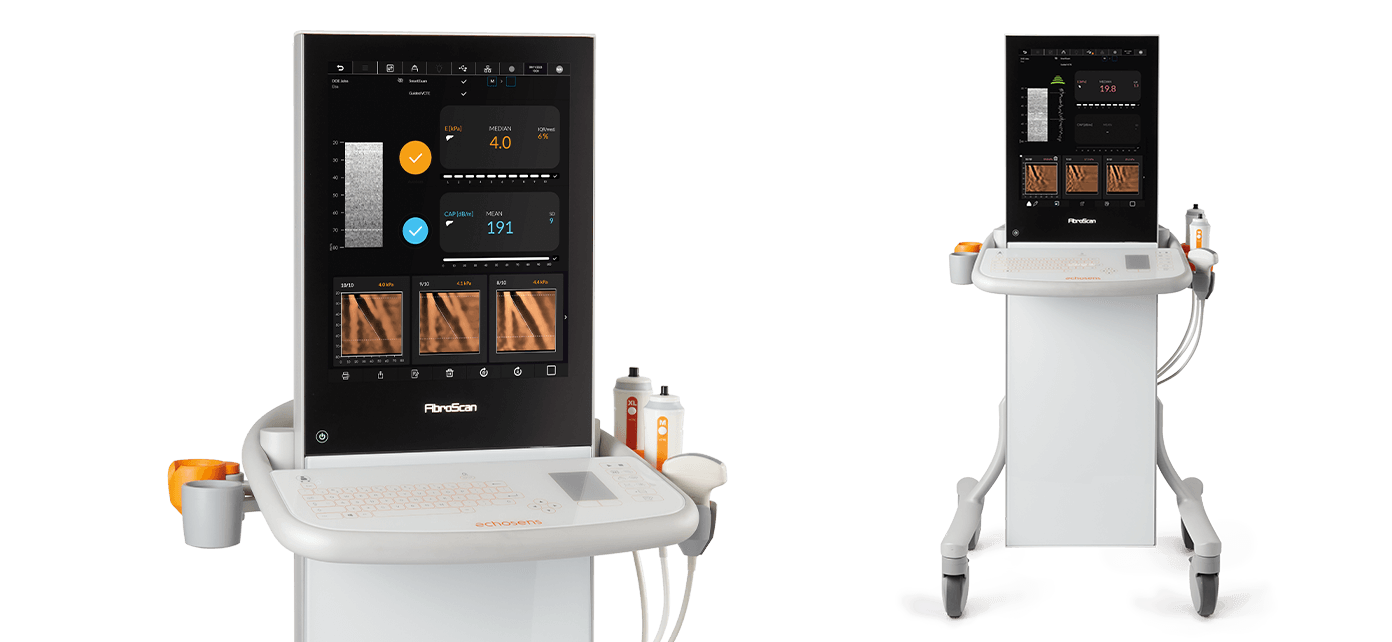
FibroScan® Expert 630 offers a high-speed processing, 19-inch touchscreen and a washable touch keyboard.
For the spleen examination with SSM by VCTE™, the M+ probe automatically adjusts to 100 Hz, adapts measurement depth, and adjusts the stiffness range.
The ultrasound localization probe is a time-saving technology for easily locating the spleen and liver in complex patients and patients with obesity.
FibroScan® Expert 630 is adapted to all patient morphologies as it can be used with all probes S+, M+ and XL+. The two probe connectors allows to easily switch between probes during exam.
From installation to training and local support, we provide you with the highest quality of services.
*SSM is a marker for non-invasive evaluation of spleen stiffness which has been used in a clinical setting to assess portal hypertension and for variceal surveillance.
1.Stefanescu H, et al. A novel spleen-dedicated stiffness measurement by FibroScan improves the screening of high-risk oesophageal varices. Liver Int. 2020;40(1):175-185. doi:10.1111/liv.14228.
2.Dajti, Elton et al. “A Combined Baveno VII and Spleen Stiffness Algorithm to Improve the Noninvasive Diagnosis of Clinically Significant Portal Hypertension in Patients With Compensated Advanced Chronic Liver Disease.” The American journal of gastroenterology vol. 117,11 (2022): 1825-1833. doi:10.14309/ajg.0000000000001887.
3.Baveno VII – R. de Franchis et al. Renewing consensus in portal hypertension. Journal of hepatology vol. 76,4 (2022): 959-974. doi:10.1016/j.jhep.2021.12.022.
FibroScan® 630 Expert is a class IIa medical device according to Rule 10 of ANNEX VIII of Regulation EU 2017/745 (CE 0459) and and is manufactured by Echosens™. FibroScan® 630 Expert is intended to provide: Liver stiffness measurements at a shear wave frequency of 50 Hz, liver ultrasound attenuation measurements (CAP: Controlled Attenuation Parameter) at 3.5 MHz and spleen stiffness measurements at a shear wave frequency of 100 Hz. FibroScan® 630 Expert is a non-invasive device intended to aid clinical management, diagnosis, and monitoring of patients with confirmed or suspected chronic liver disease, as part of an overall assessment of the liver. FibroScan® device may aid the healthcare professionals in the assessment of liver fibrosis, steatosis, and in determining the likelihood of cirrhosis, and its complications. FibroScan® device is used, in conjunction with other clinical and laboratory data, during liver assessment in patients with confirmed or suspected chronic liver disease. Examinations with FibroScan® device shall be performed by an operator who has been certified by the manufacturer or its approved local representative. Operators are expressly recommended to carefully read the instructions given in the user manual and on the labelling of these products. Check cost defrayal conditions with paying bodies.

Seamless liver health assessment for all

Optimize measurement accuracy on all patient morphologies

Cloud-based solution to assist clinicians in providing comprehensive liver care.

Optimize clinical workflows with real-time secure data transmission